Design and synthesis of chiral 1,10-phenanthroline ligand, and application in palladium catalyzed asymmetric 1,4-addition reactions - ScienceDirect
![14221-01-3・Tetrakis(triphenylphosphine)palladium(0)・209-14643・203-14641[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation 14221-01-3・Tetrakis(triphenylphosphine)palladium(0)・209-14643・203-14641[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/sc/01/14221-01-3.png)
14221-01-3・Tetrakis(triphenylphosphine)palladium(0)・209-14643・203-14641[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation

Design and synthesis of chiral 1,10-phenanthroline ligand, and application in palladium catalyzed asymmetric 1,4-addition reactions - ScienceDirect

Highly active palladium catalysts containing a 1,10-phenanthroline analogue N-heterocyclic carbene for room temperature Suzuki-Miyaura coupling reactions of aryl chlorides with arylboronic acids in aqueous media - ScienceDirect

Platinum(II) and Palladium(II) complexes with 1,10-phenanthroline and pyrrolidinedithiocarbamato ligands: synthesis, DNA-binding and anti-tumor activity in leukemia K562 cell lines | Semantic Scholar

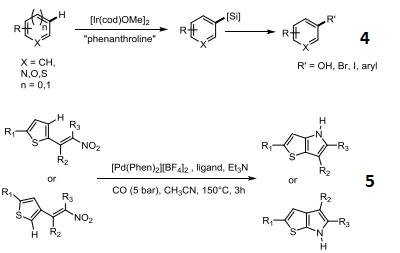
Palladium‐Catalyzed Intramolecular Cyclization of Nitroalkenes: Synthesis of Thienopyrroles - El‐Atawy - 2017 - European Journal of Organic Chemistry - Wiley Online Library

Synthesis of Indoles by Intermolecular Cyclization of Unfunctionalized Nitroarenes and Alkynes: One‐Step Synthesis of the Skeleton of Fluvastatin - Ragaini - 2009 - European Journal of Organic Chemistry - Wiley Online Library

SYNTHESIS, CHARACTERIZATION, AND CYTOTOXICITY OF MIXED-LIGAND COMPLEXES OF PALLADIUM(II) WITH 1,10-PHENANTHROLINE AND N-CARBONYL-L-ISOLEUCINE DIANION - тема научной статьи по химии из журнала "Координационная химия"

China CAS 14783-10-9 Dichloro (1, 10-phenanthroline) Palladium (II) C12h8cl2n2pd - China Palladium Catalyst, Ruthenium Catalyst

Dichloro (1,10-phenanthroline-5,6-dione) palladium (II) complex supported by mesoporous silica SBA-15 as a photocatalyst for degradation of 2,4-dichlorophenol - ScienceDirect
![PDF] X-ray structure characterization of palladium(II) ternary complexes of pyridinedicarboxylic and phthalic acid with phenanthroline and bipyridine. | Semantic Scholar PDF] X-ray structure characterization of palladium(II) ternary complexes of pyridinedicarboxylic and phthalic acid with phenanthroline and bipyridine. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/fc9bb3a11af824d13e1d63c9410c7c5569a477b7/1-Figure1-1.png)
PDF] X-ray structure characterization of palladium(II) ternary complexes of pyridinedicarboxylic and phthalic acid with phenanthroline and bipyridine. | Semantic Scholar
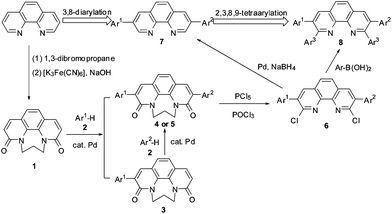
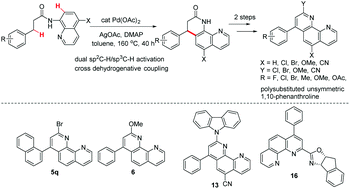
Direct arylation of phenanthroline derivatives via oxidative C–H/C–H cross-coupling: synthesis and discovery of excellent ligands - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C2OB27325J
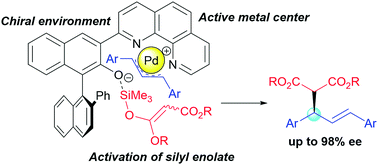
Design of bifunctional chiral phenanthroline ligand with Lewis basic site for palladium-catalyzed asymmetric allylic substitution - Chem. Commun. - X-MOL
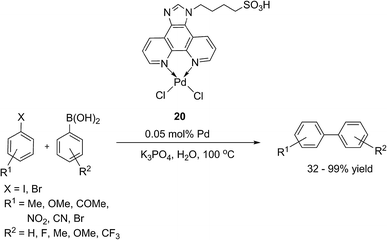
Recent Advances in the Palladium Catalyzed Suzuki–Miyaura Cross-Coupling Reaction in Water | SpringerLink

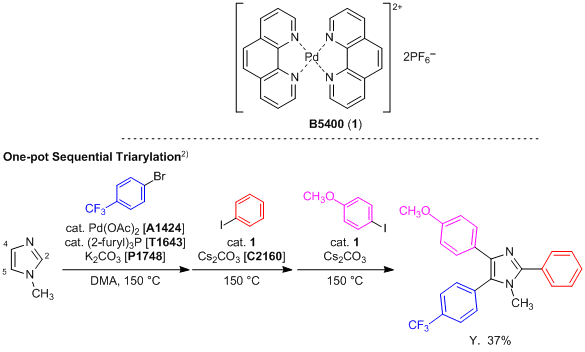
A new method for the reaction of cross-coupling: preparation of 5,5′-bi(1,10-phenanthroline) in: Heterocyclic Communications Volume 23 Issue 1 (2017)

The palladium–phenanthroline catalyzed carbonylation of nitroarenes to diarylureas: Effect of chloride and diphenylphosphinic acid - ScienceDirect
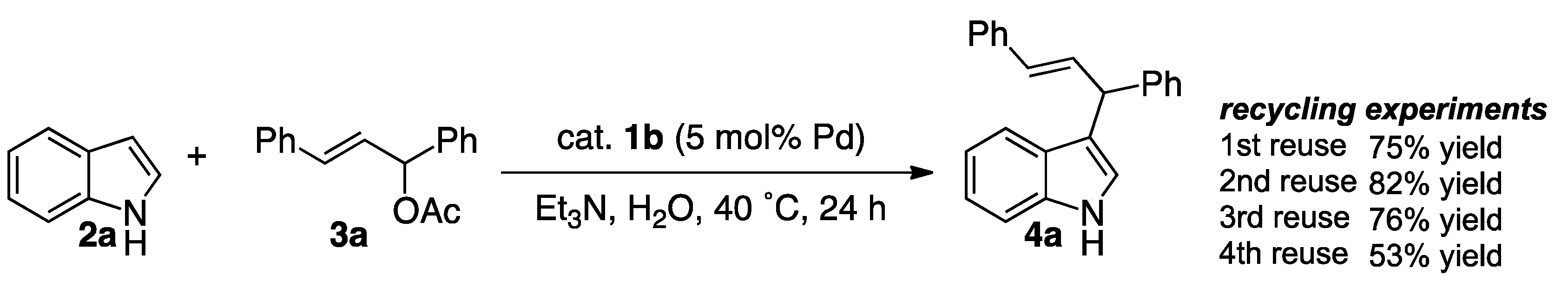
Catalysts | Free Full-Text | Friedel–Crafts-Type Alkylation of Indoles in Water Using Amphiphilic Resin-Supported 1,10-Phenanthroline–Palladium Complex under Aerobic Conditions