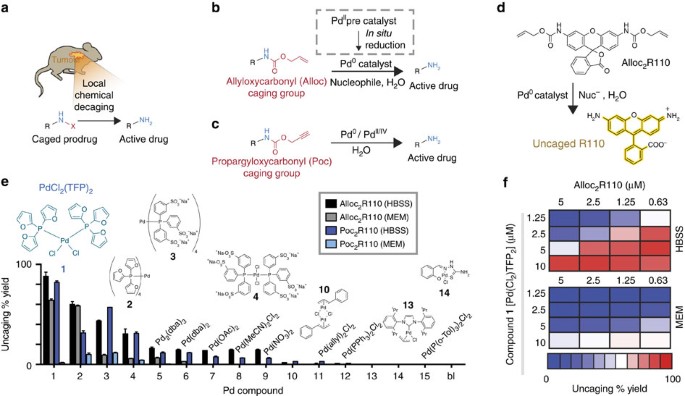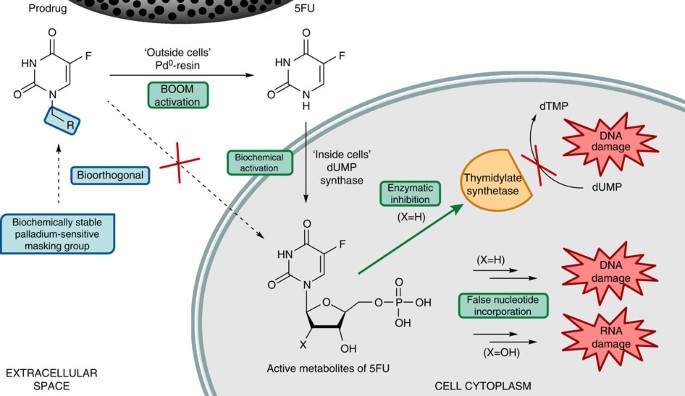
Extracellular palladium-catalysed dealkylation of 5-fluoro-1-propargyl-uracil as a bioorthogonally activated prodrug approach | Nature Communications

Recent Developments in Metal‐Catalyzed Bio‐orthogonal Reactions for Biomolecule Tagging - Jang - 2019 - ChemBioChem - Wiley Online Library

Cancer-derived exosomes loaded with ultrathin palladium nanosheets for targeted bioorthogonal catalysis. - Abstract - Europe PMC
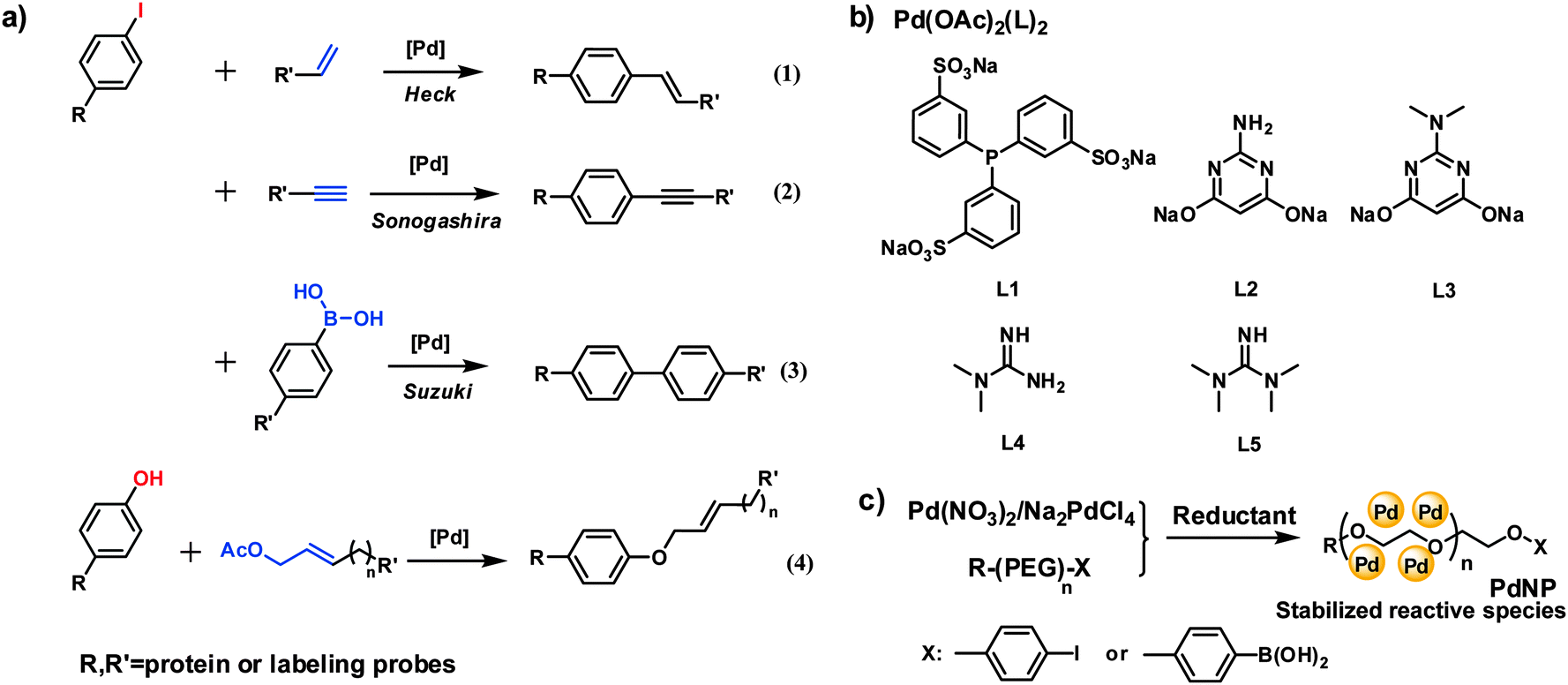
Transition metal-mediated bioorthogonal protein chemistry in living cells - Chemical Society Reviews (RSC Publishing)
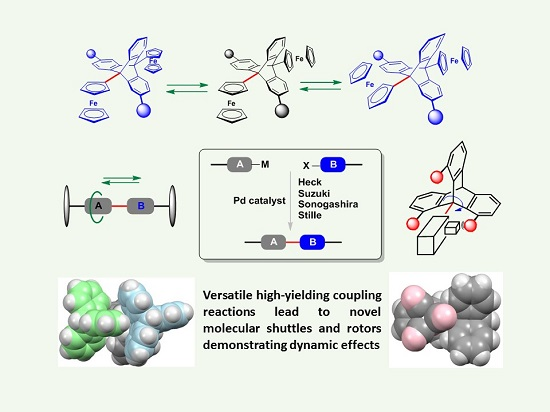
Molecules | Free Full-Text | Palladium-Catalysed Coupling Reactions En Route to Molecular Machines: Sterically Hindered Indenyl and Ferrocenyl Anthracenes and Triptycenes, and Biindenyls | HTML
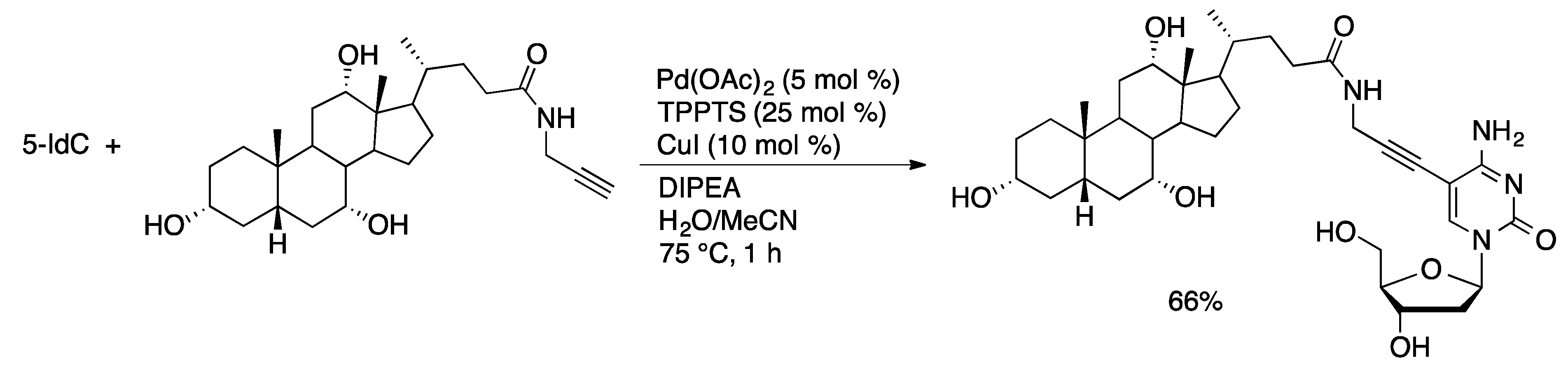
Molecules | Free Full-Text | Palladium-Catalyzed Modification of Unprotected Nucleosides, Nucleotides, and Oligonucleotides | HTML

Cancer-derived exosomes loaded with ultrathin palladium nanosheets for targeted bioorthogonal catalysis | Nature Catalysis
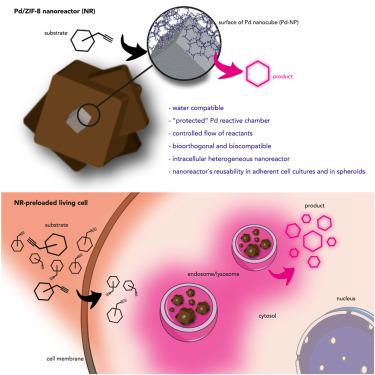
Core-Shell Palladium/MOF Platforms as Diffusion-Controlled Nanoreactors in Living Cells and Tissue Models. - Cell Rep. Phys. Sci. - X-MOL

Synthesis of d-glyco-alkynone derivatives via carbonylative Sonogashira reaction† - RSC Adv. - X-MOL
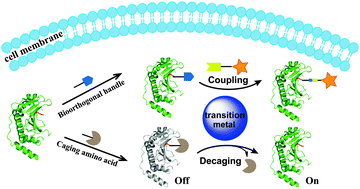
Transition metal-mediated bioorthogonal protein chemistry in living cells - Chemical Society Reviews (RSC Publishing)

a) Suzuki–Miyaura cross-coupling reactions between genetically encoded... | Download Scientific Diagram

Molecules | Free Full-Text | Palladium-Catalyzed Modification of Unprotected Nucleosides, Nucleotides, and Oligonucleotides | HTML
![PDF] Development and Bioorthogonal Activation of Palladium-Labile Prodrugs of Gemcitabine | Semantic Scholar PDF] Development and Bioorthogonal Activation of Palladium-Labile Prodrugs of Gemcitabine | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7463568d3d77326fd67d807417cdb2dda2f80ba6/3-Figure1-1.png)
PDF] Development and Bioorthogonal Activation of Palladium-Labile Prodrugs of Gemcitabine | Semantic Scholar
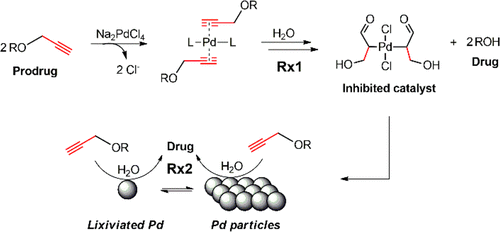
Mechanism of Palladium(II)-Mediated Uncaging Reactions of Propargylic Substrates,ACS Catalysis - X-MOL
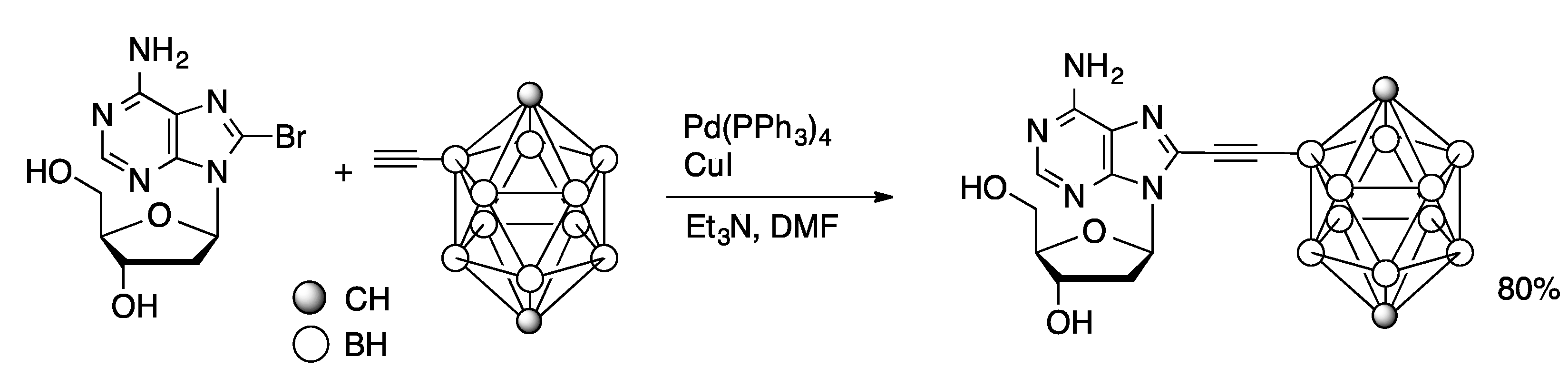
Molecules | Free Full-Text | Palladium-Catalyzed Modification of Unprotected Nucleosides, Nucleotides, and Oligonucleotides | HTML

Recent advances in bioorthogonal reactions for site-specific protein labeling and engineering - ScienceDirect