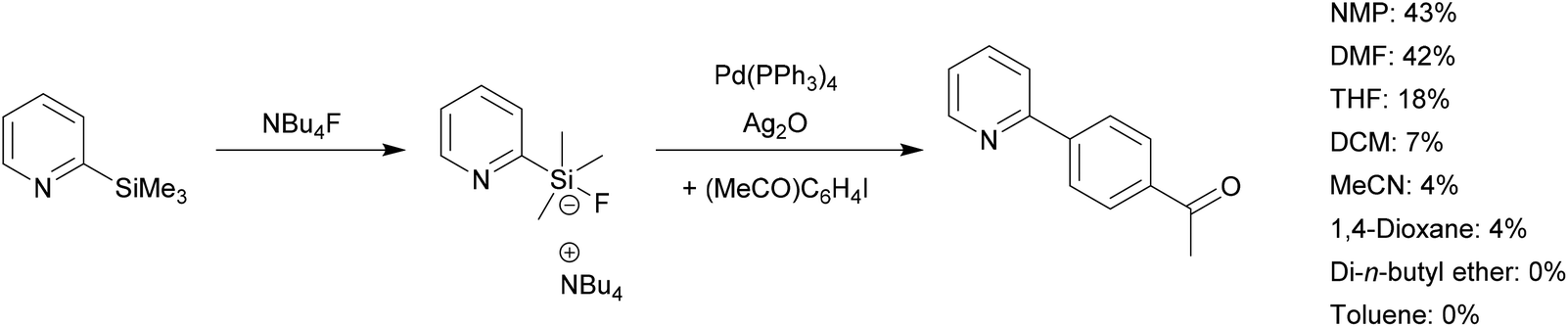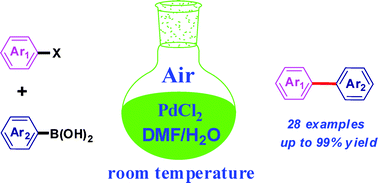
A simple and efficient protocol for a palladium-catalyzed ligand-free Suzuki reaction at room temperature in aqueous DMF - Green Chemistry (RSC Publishing)
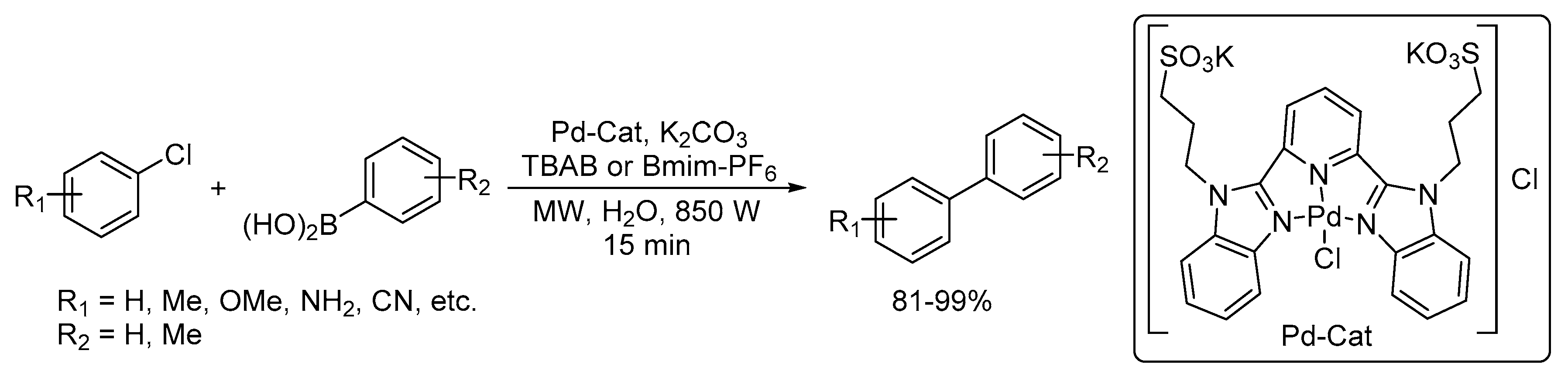
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

Combination of a Suzuki cross-coupling reaction using a water-soluble palladium catalyst with an asymmetric enzymatic reduction towards a one-pot process in aqueous medium at room temperature - ScienceDirect

Stereoselectivity in Pd-catalysed cross-coupling reactions of enantioenriched nucleophiles | Nature Reviews Chemistry

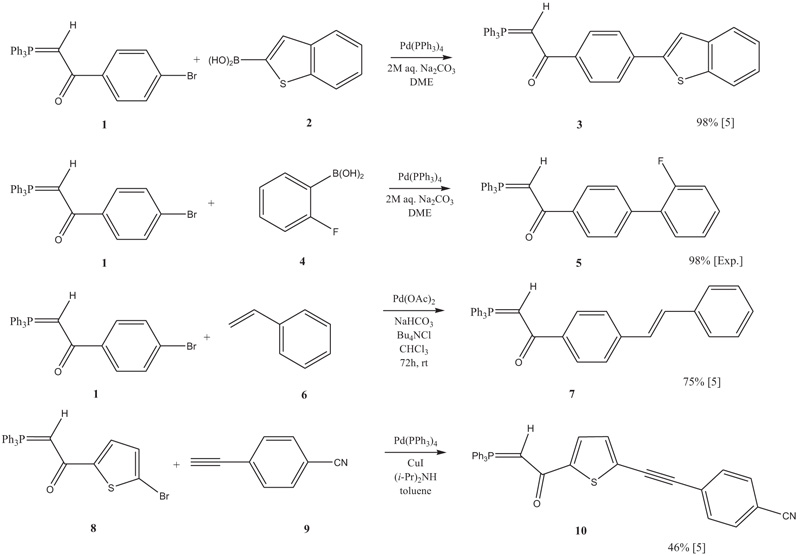
Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
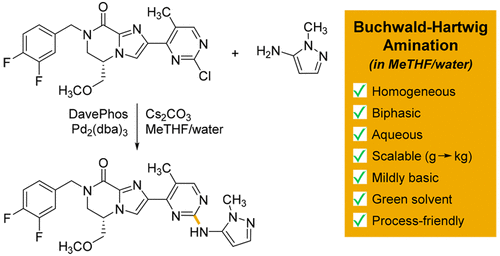
Biphasic Aqueous Reaction Conditions for Process-Friendly Palladium- Catalyzed C–N Cross-Coupling of Aryl Amines - Org. Process Res. Dev. - X-MOL
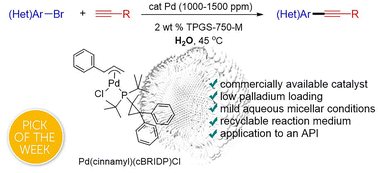
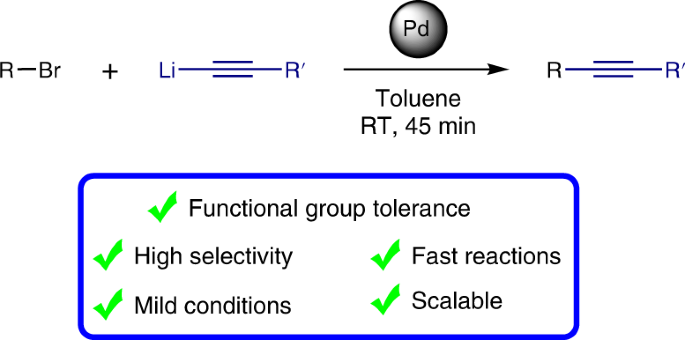
ppm Pd-catalyzed, Cu-free Sonogashira couplings in water using commercially available catalyst precursors - Chemical Science (RSC Publishing)

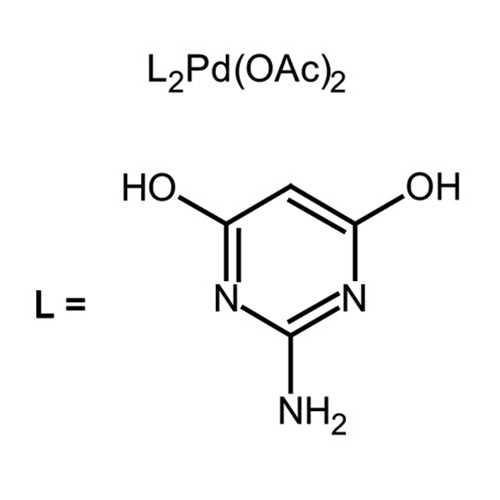
Table 1 from A Simple Hydrophilic Palladium(II) Complex as a Highly Efficient Catalyst for Room Temperature Aerobic Suzuki Coupling Reactions in Aqueous Media | Semantic Scholar

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
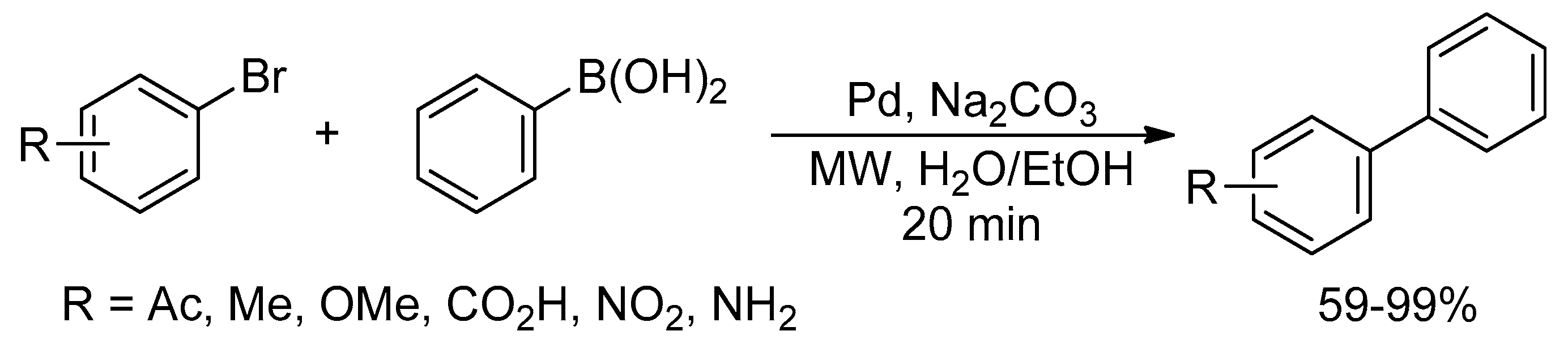
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Simple aminobenzoic acid promoted palladium catalyzed room temperature Suzuki–Miyaura cross-coupling reaction in aqueous media - ScienceDirect
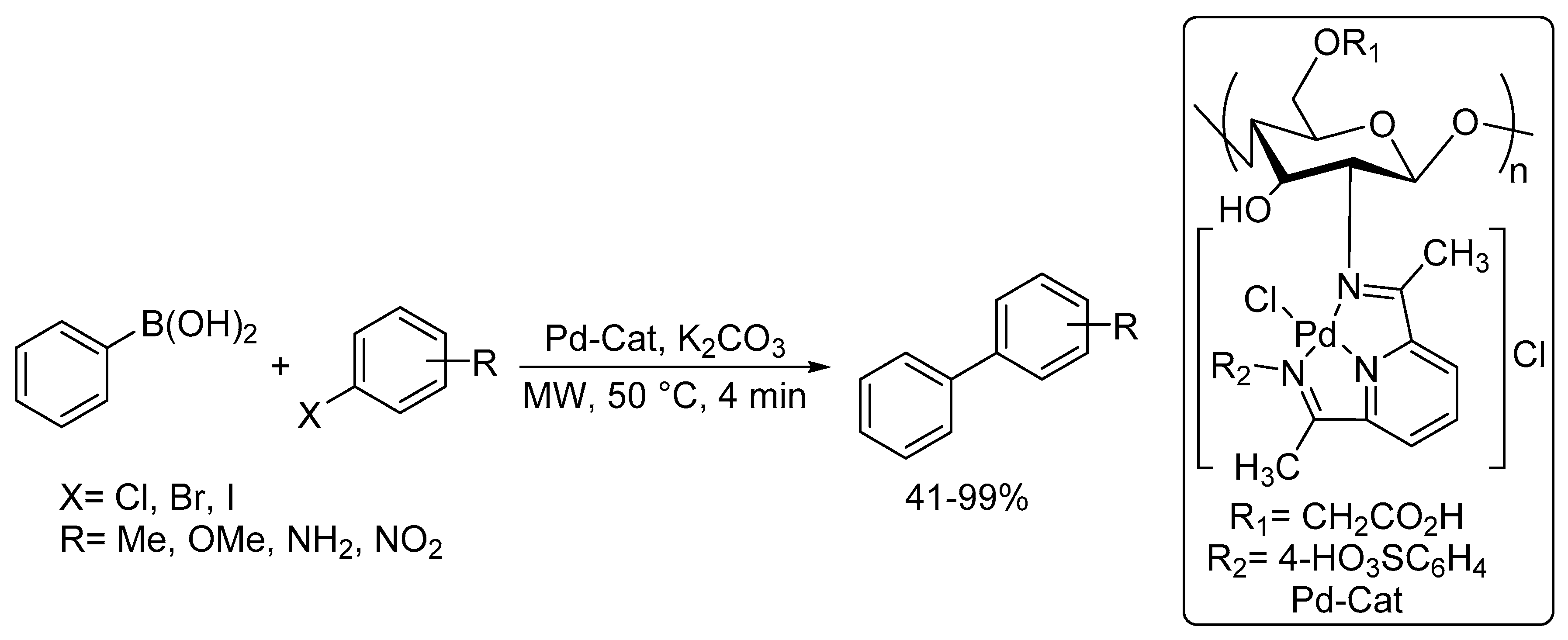
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML